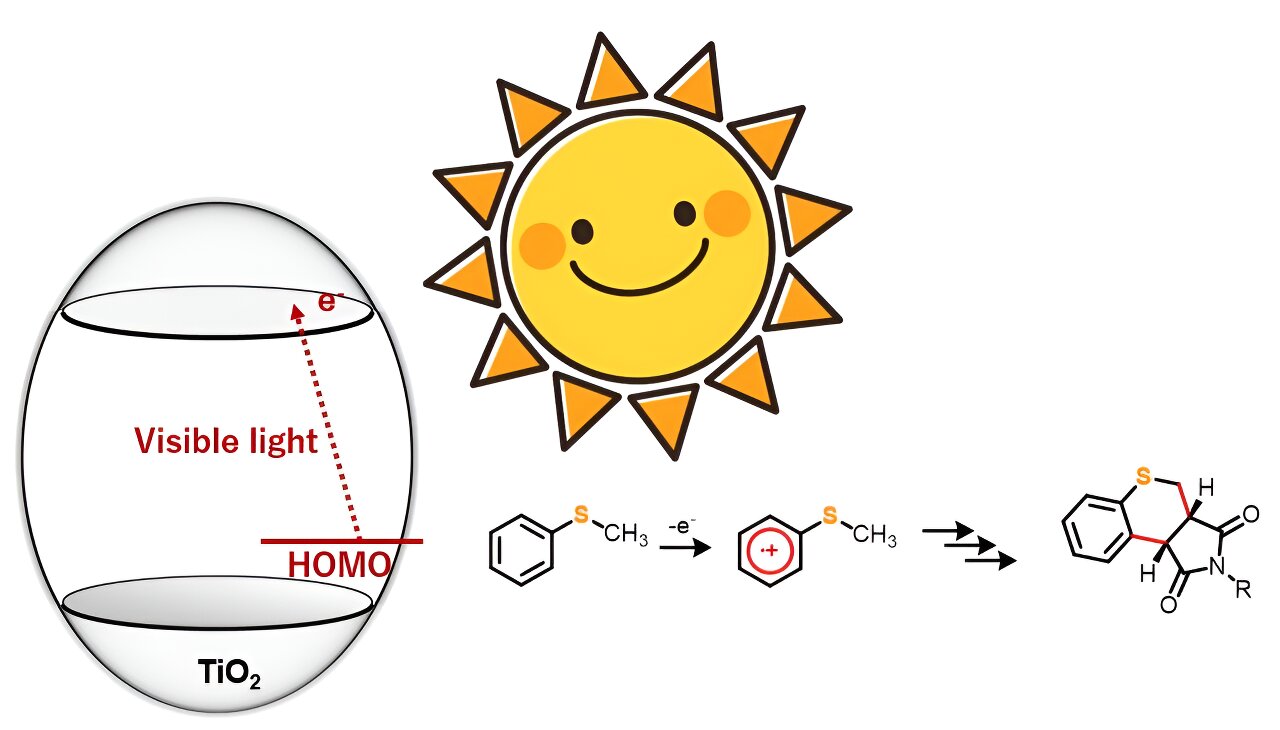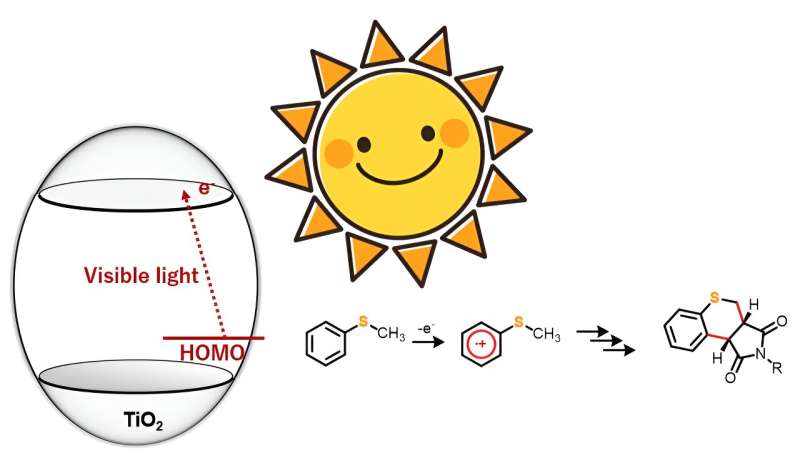
Heterocyclic compounds are natural molecules with a ring composition comprising at least two or additional components. In most instances, these rings are composed of carbon atoms along with one or a lot more other factors these as nitrogen, oxygen, or sulfur. They are really sought after as raw elements in the chemical and pharmaceutical field, owing to their versatility and great physiological routines.
Even though various solutions are available for synthesizing these compounds, most of them involve substantial temperature and pressure problems, or the use of precious steel catalysts, adding to the financial and environmental charge of manufacturing heterocyclic organic compounds.
Now, nonetheless, a workforce of scientists from Japan and Bangladesh have proposed a easy however productive approach for conquering these problems. Their research was recently published in the journal Advanced Synthesis & Catalysis. Working with the proposed method, the group demonstrated the synthesis of 20 sulfur-containing heterocyclic compounds in the existence of photocatalyst titanium dioxide (TiO2) and noticeable light-weight.
The review was led by Professor Yutaka Hitomi from the Section of Used Chemistry, Graduate Faculty of Science and Engineering, Doshisha University, and co-authored by a Ph.D. applicant Pijush Kanti Roy from Doshisha University, Associate Professor Sayuri Okunaka from Tokyo Town College, and Dr. Hiromasa Tokudome from Analysis Institute, TOTO Ltd.
TiO2 as a photocatalyst for driving organic reactions has captured the notice of synthetic chemists for a although now. Having said that, several this kind of processes require ultraviolet gentle to result in the response. In this research, even so, the exploration staff identified that below anaerobic disorders, sulfur-containing organic and natural compounds like thioanisole derivatives, when hit with blue mild, reacted with maleimide derivatives to kind twin carbon–carbon bonds, yielding a new heterocyclic natural and organic compound.
“We noticed that although ultraviolet gentle generates extremely oxidative holes, our solution enables for the selective one particular-electron oxidation of the substrate molecules making use of visible light. This tactic can consequently be employed in numerous natural chemical reactions,” explains Prof. Hitomi.
The researchers chose five 4-substituted thioanisoles and four N-substituted maleimides for the annulation or ring formation reactions. The crew irradiated the starting off materials with blue gentle (wavelength > 420 nm) but observed no response. However, introducing TiO2 into the reaction technique led to the synthesis of 20 diverse thiochromenopyrroledione derivatives with reasonable-to-large produce. They found that in just 12 several hours of publicity to blue light-weight, the reaction amongst thioanisole and N-benzylmaleimide led to the formation of a thiochromenopyrroledione spinoff with 43% produce, which was shut to the theoretical most yield of 50%.
The investigate staff also observed substituent impact in the reactions to comprehend the corresponding mechanistic aspects. From the final results, they postulated that the reaction proceeds through charge transfer from thioanisole to the conduction band of TiO2. Furthermore, they advised that irradiation with blue gentle induced a person-electron oxidation of thioanisole, which further more initiated the generation of α-thioalkyl radicals as a result of deprotonation.
In summary, this new and refined solution demonstrates the opportunity of TiO2 for obvious light photocatalysis for organic and natural synthesis. It also provided important insights into the chemistry of advanced heterocyclic compound synthesis. Shifting ahead, this tactic can open up new options for transitioning from recent source-intensive industrial chemical processes to a additional electrical power-economical system.
Prof. Hitomi claims, “What drove our examine was the desire to support in the advancement of a sustainable chemical sector, and our findings look to be a positive phase in this direction.”
“We think that the widespread adoption of this visible gentle-driven technologies could help in obtainable and very affordable synthesis of prescription drugs, with its profound impacts on the overall health and well-getting of tens of millions of individuals around the world.” Thanks to the endeavours of Prof. Hitomi and his workforce, their study has opened up new avenues in the subject of organic synthesis, with the likely to revolutionize numerous chemical industries.
Extra info:
Pijush Kanti Roy et al, Blue Light‐Promoted Synthesis of Thiochromenopyrroledione Derivatives via Titanium Dioxide‐Catalyzed Dual Carbon–Carbon Bond Formation with Thioanisole and Maleimide Derivatives, Superior Synthesis & Catalysis (2023). DOI: 10.1002/adsc.202301021
Quotation:
Chemical synthesis employing titanium dioxide: An eco-friendly and modern technique (2024, January 1)
retrieved 13 January 2024
from https://phys.org/information/2024-01-chemical-synthesis-titanium-dioxide-eco-helpful.html
This document is subject to copyright. Apart from any truthful dealing for the goal of non-public examine or research, no
element may be reproduced devoid of the composed permission. The content material is supplied for details purposes only.